Integrated Study Dashboard
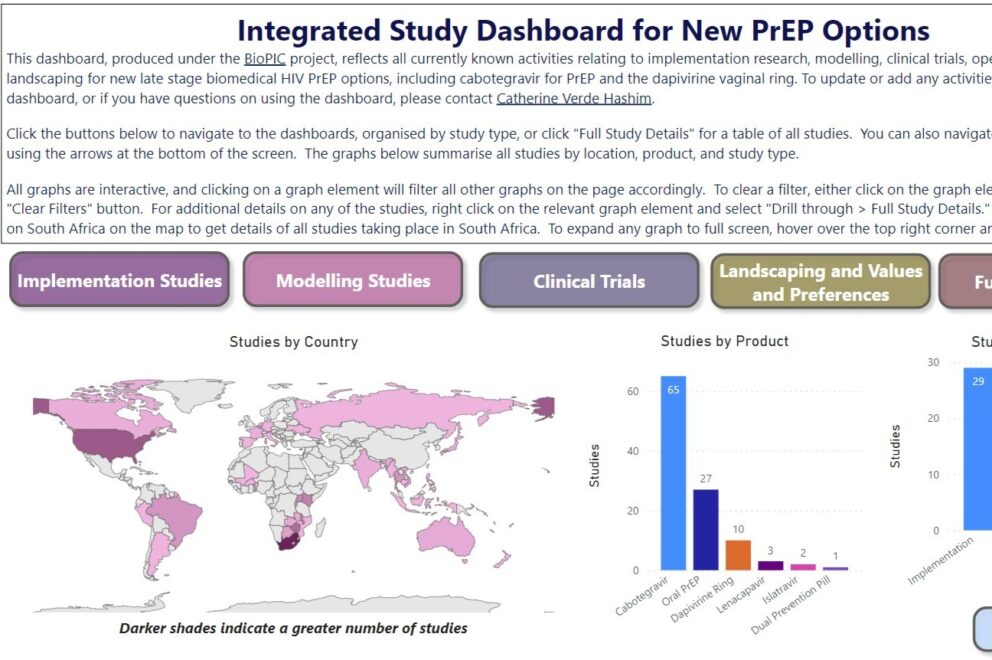
This dashboard, produced under the BioPIC project, reflects all currently known activities relating to implementation research, modelling, clinical research, and landscaping for new late stage biomedical HIV PrEP options, including cabotegravir for PrEP and the dapivirine vaginal ring. To update or add any activities missing from the dashboard, or if you have questions on using the dashboard, please contact Catherine Verde Hashim to ensure they are included in the next update.
As a collaborative platform, BioPIC supports introduction, planning, and information sharing for the successful introduction of HIV prevention products. One of BioPIC’s key roles is to serve as a clearinghouse to monitor ongoing and planned activities related to HIV prevention and product introduction with a particular focus on injectable cabotegravir for PrEP. The goal is to facilitate a shared understanding of the various partners, funders, location, scope, and focus of various implementation research and projects.