Products
PrEP works. Investment in more options must continue.
Products
The development of PrEP products as interventions in HIV prevention represents one of the greatest achievements in the global response to HIV. If combined with community-centered approaches to delivering all HIV prevention options, PrEP products will be instrumental to ending the epidemic. Key to that potential is developing an array of products that can meet the diverse needs of people who need HIV prevention.
The list of now approved products is growing. The list of products in development also needs continued investment so everyone who needs to protect themselves finds a tool that works for them. For an overview of the product registration process, click here.
Approved
- Oral PrEP
- The dapivirine vaginal ring for PrEP (DAP Ring, DVR, or PrEP Ring)
- Injectable cabotegravir for PrEP (Injectable CAB)
In Development
Additional strategies in development, including multipurpose prevention products, could protect against HIV, STIs and pregnancy. More primary prevention tools mean more choice, and history shows that more choice increases the use of all products. Learn more about the status of tomorrow’s strategies here.
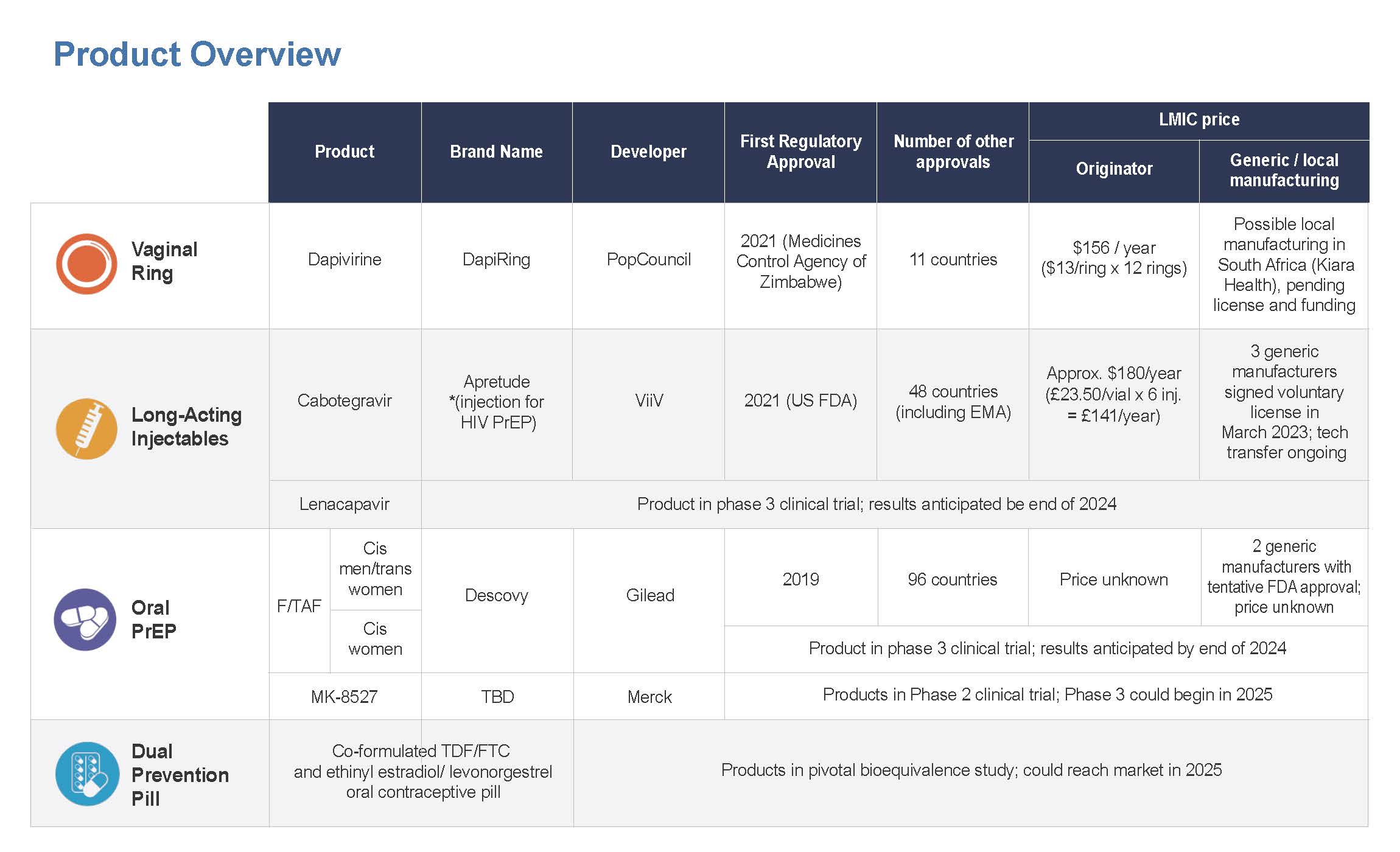
The graphic below provides an overview of PrEP products currently available and in late-stage clinical trials. Click the image to enlarge.